Fluorine Notes, 2000, 8, 7-8
Synthesis and application of  -bromo-perfluoroalkylvinyl ethers.
-bromo-perfluoroalkylvinyl ethers.
Chekmariov P.M., Andrushin V.M., N.A. Dreyman
Report IV. Synthesis of new fluoropolymers by interaction of fluoroolefins with  -bromo-perfluoroalkylvinyl ethers and their derivatives.
-bromo-perfluoroalkylvinyl ethers and their derivatives.
Introduction
As it was reported earlier (1),  -bromo-perfluoroalkylvinyl ethers (BrAVE) can be used to modify polymers based on fluorine-containing olefins because operational functionality of fluoroplasts made from fluoro-substituted olefins of conventional structures have been exhausted in general and development of technique requires new fluorine-containing polymers possessing an improved set of properties providing long term efficiency under extreme conditions.
-bromo-perfluoroalkylvinyl ethers (BrAVE) can be used to modify polymers based on fluorine-containing olefins because operational functionality of fluoroplasts made from fluoro-substituted olefins of conventional structures have been exhausted in general and development of technique requires new fluorine-containing polymers possessing an improved set of properties providing long term efficiency under extreme conditions.
One of the routes to create new fluoropolymers of new generation is the use of new fluoromonomers which allow to create cross-linked polymer structures providing an increase in hardness, heat and chemical resistance, a decrease in the swelling factor etc.
Another route to create new fluoropolymers is development of perfluorinated monomers with functional groups able to different polymer-analogous conversions. In this case the polymeric chain serves like a carrier of reaction functional centers. This makes prospects for creation of polymers for very new areas of application, for example to give hydrophily to hydrophobic materials, to synthesize cross-linked polymers, ion-conductive materials etc.(2)
The proposed report presents the results of the research on copolymerization of  -bromo-perfluoroalkylvinyl ethers and
-bromo-perfluoroalkylvinyl ethers and  -bromomethyl perfluoroalkylvinyl ethers (BMAVE) on their base and perfluoroalkylendivinyl ethers (DVE) with the most common fluoroolefins, tetrafluoroethylene (TFE), and vinyliden fluoride (VDF).
-bromomethyl perfluoroalkylvinyl ethers (BMAVE) on their base and perfluoroalkylendivinyl ethers (DVE) with the most common fluoroolefins, tetrafluoroethylene (TFE), and vinyliden fluoride (VDF).
1. Copolymers based on fluoroolefins and 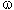 -bromo-perfluoroalkylvinyl ethers.
-bromo-perfluoroalkylvinyl ethers.
1.1.Copolimerization of  -bromo-perfluoroalkylvinyl ethers with fluoroolefins.
-bromo-perfluoroalkylvinyl ethers with fluoroolefins.
Analysis of methods to produce different fluoropolymers based on tetrafluoroethylene and vinyliden fluoride has shown that the copolymerization is carried out in general in two ways: either in a medium of organofluoric solvents, chladonespreferably, or by a water-emulsion method (3,4).
The reaction was carried out in a special polymerization reactor (0.2-2.0 dm3) at a temperature of 35-65oC and a pressure of from 3 to 14 kg/cm2 in an inert oxygen-free medium. Ammonia persulfate (PSA) was used as an initiator in the water-emulsion process and bis(perfluorocyclohexanonium) peroxide (DAP-5) was used for the same purpose in a medium of R-113. A compensation method was used to study the copolymerization process. It consisted in a periodic feed of the most active (consumable) monomer to the reactor to provide the constancy of the monomers ratio during the course of the reaction. In our case the pressure in the reactor was held at the necessary level by feeding tetrafluoroethylene or vinyliden fluoride from a special calibrated vessel because partial pressures of other components of the reaction mixture are negligible compared with them.
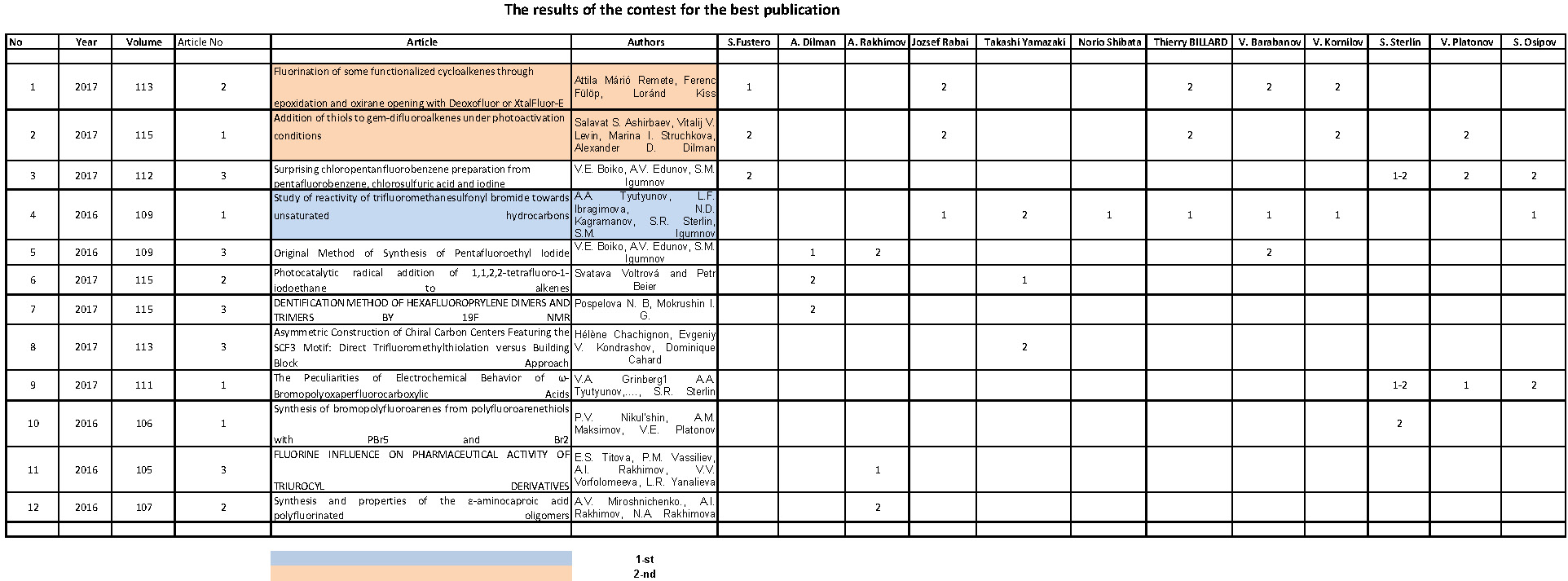
Table 1
PTR -the fluidity of solution.
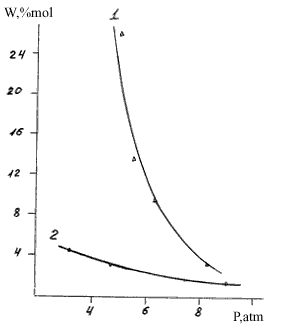
Fig.1. Dependence of the copolymer of TFE and  -bromo -perfluoroalkylvinyl ethers composition (W,mol %) on the TFE pressure (P,kg/cm2) in a water-emulsion medium
-bromo -perfluoroalkylvinyl ethers composition (W,mol %) on the TFE pressure (P,kg/cm2) in a water-emulsion medium
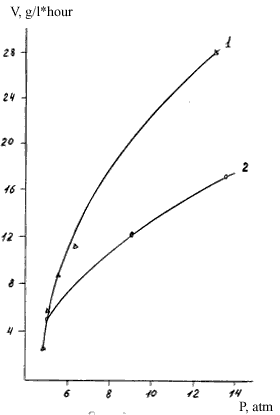
Fig.2. Dependence of the copolomerization rate of TFE and  -bromo -perfluoroalkylvinyl ethers in a water-emulsion medium (V,g/L*h) on the TFE pressure (P,kg/cm2)
-bromo -perfluoroalkylvinyl ethers in a water-emulsion medium (V,g/L*h) on the TFE pressure (P,kg/cm2)
Evidently, it may be explained by the chladone participation in the chain transfer reaction to form low-activity radicals.
As it is seen in Fig.1 and 2, the copolymerization rate and the  -bromo -perfluoroalkylvinyl ethers content in the copolymer are strongly dependent on the TFE concentration ( pressure in the reactor) and increasing the length of perfluoroalkylen fragment in the
-bromo -perfluoroalkylvinyl ethers content in the copolymer are strongly dependent on the TFE concentration ( pressure in the reactor) and increasing the length of perfluoroalkylen fragment in the  -bromo -perfluoroalkylvinyl ethers molecule causes a drastic drop in its reactivity.
-bromo -perfluoroalkylvinyl ethers molecule causes a drastic drop in its reactivity.
A character of dependencies of the copolymerization reaction of vinyliden fluoride with  -bromo -perfluoroalkylvinyl ethers in a whole is the similar as that of the copolymerization of TFE with
-bromo -perfluoroalkylvinyl ethers in a whole is the similar as that of the copolymerization of TFE with  -bromo -perfluoroalkylvinyl ethers.
-bromo -perfluoroalkylvinyl ethers.
An increase of the pressure of the main monomer results in an abrupt increase in the reaction rate while the  -bromo -perfluoroalkylvinyl ethers content in the copolymer drops in the same drastic way.
-bromo -perfluoroalkylvinyl ethers content in the copolymer drops in the same drastic way.
1.2. Investigation of conditions of cross-linking of fluorocopolymers based on 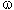 -bromo -perfluoroalkylvinyl ethers.
-bromo -perfluoroalkylvinyl ethers.
Efforts on cross-linking the copolymers produced according to the radical mechanism in the presence of perfluorodiacyl peroxide and triallylisocyanurate ( similarly to bromic fluorocarbons) did not give a positive result due to a low mobility of fluoropolymer molecules at a cross-linking temperature (60-80oC) which is much lower than the melting point (230-250oC) of the fluoropolymer.
As the critical reaction stage for the polymer analogous reaction is diffusion of reagents interacting with the functional copolymer group, the investigation of cross-linking conditions for the polymer chains to bromine-containing groups was carried out using soluble fluoropolymers based on vinyliden fluoride and  -bromo -perfluoroalkylvinyl ethers. Hexafluorodiphenylolpropane (DFP) and triallylisocyanurate were used as a cross-linking agent (5-7), curing agent (8) to produce thermostable polymers possessing thermooxidizing and thermal and chemical stability (9-12).
-bromo -perfluoroalkylvinyl ethers. Hexafluorodiphenylolpropane (DFP) and triallylisocyanurate were used as a cross-linking agent (5-7), curing agent (8) to produce thermostable polymers possessing thermooxidizing and thermal and chemical stability (9-12).
To produce cross-linked fluoropolymer, the copolymer of vinyliden fluoride and 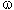 -bromo -perfluoroalkylvinyl ethers was dissolved in an acetone-water solution and cross-linked in a water solution with sodium hexafluoropropanediphenolate in amount of 3 wt. % from the total amount of the reagent, water was then added to the solution at vigorous stirring to the polymer precpitation. The product produced was separated from water and consequently dried first at 100oC for 3 hours, then at 190oC for 3 hours, the polymer was then washed with water and dried.
-bromo -perfluoroalkylvinyl ethers was dissolved in an acetone-water solution and cross-linked in a water solution with sodium hexafluoropropanediphenolate in amount of 3 wt. % from the total amount of the reagent, water was then added to the solution at vigorous stirring to the polymer precpitation. The product produced was separated from water and consequently dried first at 100oC for 3 hours, then at 190oC for 3 hours, the polymer was then washed with water and dried.
The polymer produced was no longer soluble in acetone that was an evidence of polymer cross-linking as a result of DFP difunctionality and copolymer polyfunctionality .
As soon as the copolymer of TFE with  -bromo -perfluoroalkylvinyl ethers is insoluble in acetone, the modification method was different: an acetone-water solution of sodium hexafluoropropanediphenolate was added into the copolymer swollen in acetone, the mixture was kept for a day at room temperature and then dried according to the method described above.
-bromo -perfluoroalkylvinyl ethers is insoluble in acetone, the modification method was different: an acetone-water solution of sodium hexafluoropropanediphenolate was added into the copolymer swollen in acetone, the mixture was kept for a day at room temperature and then dried according to the method described above.
Infusible copolymers are formed when not less than 3 wt% of sodium hexafluoropropanediphenolate is added. The cross-linking degree of polymers based on TFE under the mentioned conditions is lower than that of vinyliden fluoride due to low diffusion of cross-linking agent to the structure of the polymer swollen.
Triallylisocyanurate (TAIC) has been also known in literature as a cross-linking agent in processes of peroxide vulcanization of bromine-containing rubbers (13).
We studied the process of cross-linking a soluble copolymer of vinyliden fluoride and  -bromo -perfluoroalkylvinyl ethers using triallylisocyanurate and dicumenyl peroxide.
-bromo -perfluoroalkylvinyl ethers using triallylisocyanurate and dicumenyl peroxide.
The copolymer was dissolved in acetone, acetone solutions of dicumenyl peroxide and triallylisocyanurate were added and mixed carefully. After acetone distillation, the reaction mixture was kept for 4 hours at 150oC. There was observed a 18-fold decrease in the PTR (?) value of the copolymer modified in comparison with the initial one.
As a result, samples of infusible insoluble copolymers were produced, they may be applied in harsh environment (acids, alkalis, solvents, etc.) at elevated temperatures. The results of the investigation on properties of such copolymers will be stated in future reports of this series.
1.3. Synthesis of copolymers based on fluoroolefins and perfluorodienes.
The use of perfluorodienes as modifiers for fluoropolymers to improve their physical-chemical and physical-mechanical characteristics has been widely known. Perfluorinated divinyl ethers are of particular interest due to their structure features: bifunctionality of the molecules ( the presence of two olefin fragments of the same activity) allows to produce cross-linked polymers with a reduced stress fluidity and elevated heat resistance while the presence of two hetero-atoms in the comonomer chain gives specific properties to the finished product.
The cross-linking may be carried out both in the polymerization reaction and in the process of thermal processing the material.
Both methods have been studied using perfluorobutylendivinyl ether produced from w-bromotetraethylvinyl ether by interaction with active copper:
BrCF2CF2OCF=CF2  CF2=CFO(CF2) 4OCF=CF2
CF2=CFO(CF2) 4OCF=CF2
It has been determined that the introduction of less than 0.1wt% of diene into the chain of the copolymer of TFE and perfluoroalkylendivinyl ethers does not practically affect the copolymer properties while the introduction of 0.2-0.3 wt % results in approximately two-fold decrease in PTR(?), that is the evidence of the increase in the molecular mass due to the cross-linking during the copolymerization process ( the following heat treatment does not practically affect PTR?). The introduction of 0.5-0.1 wt% of diene gives materials with a very low fluidity , the following heat treatment (pressing, extrusion, film formation) results in the final cross-linking of the polymer with formation of three-dimensional structure and complete absence of fluidity.
The IR-spectroscopy data also confirm the total disappearance of the C=C bonds.
Copolymers of perfluoroalkylendivinyl ethers and vinyliden fluoride containing 1.0-1.3 wt.% of diene are high-melting soluble products, after heat treatment they completely lose their solubility and fluidity. This is an evidence that partial cross-linking proceeds during pressing at 200-250oC.
As it is seen from the data obtained, perfluoroalkylendivinyl ethers are effective modifiers for fluoroplasts that allows to produce polymers with improved performance attributes.
1.4. Production of fluoropolymers with anion-active properties.
Earlier it was briefly reported(14) about synthesis of polymers possessing anion-active properties by copolymerization of TFE with fluorinated vinyl ethers of the formula
which were produced by interaction of the appropriate  -bromo -perfluoroalkylvinyl ethers with vinyliden fluoride.
-bromo -perfluoroalkylvinyl ethers with vinyliden fluoride.
Fluorinated vinyl ethers monomers (as are 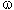 -bromo -perfluoroalkylvinyl ethers) show high activity in radical co-polymerization with TFE in R-113. The functional monomer content in the co-polymer produced attained 15 mol %. Film samples of 150-200mkm thickness were produced from fluorinated vinyl ethers monomers by a hot pressing method at a temperature of 250oC and loading up to 200kg/cm2. It is significant that there is no cross-linking to bromine, that allows further to use the end bromomethyl fragment for its polymer analogous conversions to the goal ammonium-iodine group.
-bromo -perfluoroalkylvinyl ethers) show high activity in radical co-polymerization with TFE in R-113. The functional monomer content in the co-polymer produced attained 15 mol %. Film samples of 150-200mkm thickness were produced from fluorinated vinyl ethers monomers by a hot pressing method at a temperature of 250oC and loading up to 200kg/cm2. It is significant that there is no cross-linking to bromine, that allows further to use the end bromomethyl fragment for its polymer analogous conversions to the goal ammonium-iodine group.
Amination in the side chain of the polymer was carried out in an autoclave at 50-140oC for 27-160 hours using triethylamine, diethylamine and ethylenediamine. The reaction does not practically proceed up to 100oC ( the substitution degree did not exceed 3%), at heating to 120-140oC 60% of bromine atoms are gradually substituted. Here the formation of new C=C bonds has been observed, that may be explained by the following side reaction of dehydrofluorination:
-CF2–CH2Br + HN(Alk) 2  -CF=CHBr + H N(Alk) 2F ,
-CF=CHBr + H N(Alk) 2F ,
but by a proper choice of the amination conditions this reaction can be suppressed.
Quaternization of amino-groups was carried out in a methanol medium at a temperature of 50-60oC for 50-70 hours according to the reaction:
Rf-CF2-CH2 N(Alk) 2 + IAlk  Rf-CF2N+(Alk)3 I
Rf-CF2N+(Alk)3 I
In case of need, iodine can be easily substituted for another halogen at almost the same conditions.
The copolymer produced was successfully tested in an electromeric cell. The current efficiency (without optimization of conditions) was 65%.
Conclusion.
On the basis of the copolimerization of fluoroolefins with  -bromo -perfluoroalkylvinyl ethers, new fluorinated polymers with improved performance attributes have been developed .
-bromo -perfluoroalkylvinyl ethers, new fluorinated polymers with improved performance attributes have been developed .
References
- V.M.Andrushin,N.B.Pavlova “Synthesis and application of
 -bromo-perfluoroalkylvinyl ethers”, Report1, Fluorine Notes, N5,1999
-bromo-perfluoroalkylvinyl ethers”, Report1, Fluorine Notes, N5,1999 - M.Fedtke “Chemical reactions of polymers” M., Himiya, 1990
- News in development of ion-exchange fluoropolymer membranes” Review information. NIITEHIM,M.,1985
- News in development of fluoroelastomers” Review information. NIITEHIM,M.,1983
- US patent 4663395,1985
- US patent 4673715,1986
- application for patent, Japan, 62-138547,1985
- application for patent, Germany,3624815,1986
- US patent 4695655,1984
- US patent 4649207,1986
- application for patent, Japan, 61-238761,1985
- US patent 4521623,1984
- S.P.Novitskaya et al.”Fluoroelastomers”, M.,Himiya,1988
- V.M.Andrushin, L.M.Popova, N.B.Pavlova “Synthesis and application of
 -bromo-perfluoroalkylvinyl ethers”, Report2, Fluorine Notes N6,1999
-bromo-perfluoroalkylvinyl ethers”, Report2, Fluorine Notes N6,1999
Fluorine Notes, 2000, 8, 7-8