Fluorine Notes, 2000, 8, 9-10
THE ANALYSIS OF 1,1,2 – TRIFLUORO – 1,2 – DICHLOROETHANE THERMAL DECOMPOSITION PROCESS
A.A. Nikolaev, O.V. Rozhdestvenskaya, V.G. Barabanov
It is known that one of the methods to obtain trifluorochloroethylene (I) is 1,1,2-trifluoro-1,2-dichloroethane (I) thermal decomposition process. [1,2,3]
In the previous project we analyzed the kinetics and mechanism of the said process’s main reaction, namely the thermal dehydrochlorination of the compound (II) in a flow reactor [1]. However, this process was complicated by side reactions. Hereby we provide the results of compound (II) pyrolysis analysis in a flow reactor in the presence of aqueous vapor.
Pyrolysis has been conducted in temperature range of 973 – 1123 K at various contact intervals. Compound (II) was diluted by aqueous vapor for the decrease of contribution on the part of side reactions.
At the 1,1,2-trifluoro-1,2-dichloroethane thermal decomposition process, the following compounds have been identified by the method of chromatomass-spectrometry, namely C2F3Cl, C2F3H (III), 1,2-difluorodichloroethylene (IV), CF2ClH (V), 1,2,3,3,3,- pentafluorochloropropene (VI), 1,3,3,3 –tetrafluorodichloropropene (VII), hexafluoro-1,3-butadiene (VIII), 1,2,3,3,4,4-hexafluorodichlorocyclobutane (IX), 1,1,2,3,4,4-hexafluorodichloro-3-butene (X), CO.
The most probable channel of the compound (IV) formation should be recombination of fluorochlorocarbenes, which are produced so far as is known at the decomposition of trifluorochloroethylene [4, 5].
However the analysis of the experimental data has shown the impossibility to explain the formation of 1,2-difluorodichloroethylene only by this reaction.
It is obvious that the compound (IV) is also the result of dehydrofluorination of the basic alkane.
C2F3Cl  CF2: + CFCl :
CF2: + CFCl :
2CFCl:  C2F2Cl2
C2F2Cl2
CF2ClCFClH  C2F2Cl2 + HF
C2F2Cl2 + HF
It is apparent that the mechanism of obtaining the products (IV, VII) is determined by the reactions of difluoro- and fluorochlorocarbenes addition to trifluorochloroethylene.
At the same time, carbenes are most probable to join trifluoromethylchlorocarbene, produced at the 1,2 migration of the atom of fluorine:
CF2= CFCl  :CClCF3
:CClCF3
:CClCF3  CF2= CFCl
CF2= CFCl
CClCF3: + CFCl:  C3F4Cl2
C3F4Cl2
CClCF3: + CF2:  C3F5Cl
C3F5Cl
The structure of the compounds (VI, VII) confirms this assumption.
Olefin-carbene isomerization is described in the research paper [6]. Obtaining compounds (VI, VII) according to the radical mechanism is unlikely, since in this case the radical chlorination with the adjoining of chlorine under double bond would take place.
Compounds (IX, X), as far as is known, are formed at dimerization of trifluorochloroethylene [7, 8, 9, 10].
During the analysis of 1,1,2-trifluoro-1,2-dichloroethane thermal decomposition an apparent influence of the reactor’s walls on the speed of trifluoroethylene and hexafluoro-1,3-butadiene formation has been discovered. To verify the assumption of heterogeneous character of the aforementioned reactions, tests with various surface-volume ratio (S/V, cm-1) have been carried out (Table 1).
Table 1. The influence of the surface on the yield of some 1,1,2-trifluoro-1,2-dichloroethane pyrolysis products (temperature – 1073 K, time of contact – 0,15 sec, vapor / freon molar ratio – 3:1).
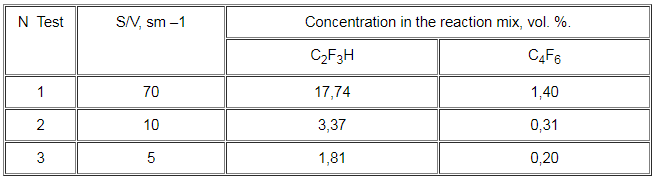
Compound (II) pyrolysis tests in the presence of the inhibitor (C3H6) and the initiator (CCl4) of the radical processes have been performed for the investigation of the said compounds’ formation.It is evident from Table 1 that the surface bears considerable influence on the speed of trifluoroethylene and hexafluoro-1,3-butadiene formation.
The absence of their influence on the speed of trifluoroethylene formation and decrease of compound (VIII) yield in the presence of propylene have been identified, which in turn confirms the radical mechanism of the said product’s formation.
Obviously, compound (VIII) is formed at the thermal dechlorination of compound (X).
Trifluoroethylene is apparently formed through the disproportionation of the basic alkane into 1,1,2-trifluorotrichloroethane, which is not thermostable at the temperature of the process, and 1,1,2-trifluoro-1--chloroethane, easily eleminating HCl to result in the formation of trifluoroethylene:
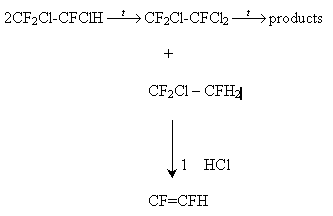
The heterogeneous character of trifluooroethylene and hexafluoro-1,3-butadiene formation allows for the exclusion of their formation reactions from the process model.
Upon the transmission to the industrial reactor, their total yield shall not exceed 0,5 - 1%.
Therefore, the process of 1,1,2-trifluoro-1,2-dichloroethane thermal decomposition may be described by the following diagram.
- C2F3Cl2H
 C2F3Cl + HCl
C2F3Cl + HCl - C2F3Cl2H
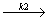 C2F2Cl2 + HF
C2F2Cl2 + HF - C2F3Cl
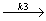 CFCl: + CF2 :
CFCl: + CF2 : - CF2 : + CFCl :
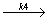 C2F3Cl
C2F3Cl - C2F3Cl + CFCl:
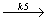 C3F4Cl2
C3F4Cl2 - C2F3Cl + CF2 :
 C3F5Cl
C3F5Cl - 2C2F3Cl
 C4F6Cl2
C4F6Cl2 - CF2 : + H2O
 CO + 2HF
CO + 2HF - CFCl: + H2O
 CO + HCl + HF
CO + HCl + HF - CF2: + HCl
 CF2ClH
CF2ClH - CF2ClH
 CF2: + HCl
CF2: + HCl - 2CFCl :
 C2F2Cl2
C2F2Cl2
For above diagram the structural and parametric identification of model was executed on base of experimental data.
The main task of parametric identification - to find the vector of kinetic constants, the elements of which are pre-exponential functions, the energies of activation:
abs (f(p) - Y) < Em
Y being the generalized vector of the experimental data, i.e. of all the experimental values of molar concentrations related to the present complex reaction;
f(p) being the generalized vector of the design data for the identified set of kinetic constants p, the latter being obtained under the same conditions as the corresponding elements of the vector Y;
Em being the vector of majorant accuracy estimates of the experiments E.
E > Sup abs (E)
The methodology of the parametric identification of the model was based on the method of least squares (MLS). The realization of the MLS for the present model being nonlinear in its parameters was performed on the ground of the Gauss-Newton method [11 ]. Elsoda method designed for the solution of the stable systems of differential equations was used as the system’s numerical integration method.[12]
The Arrhenius parameters of the speed constants of the analyzed process's stages are provided hereby in Table 2.
Table 2.The speed constants Arrhenius parameters of the 1,1,2-trifluoro-1,2-dichloroethane thermal decomposition process
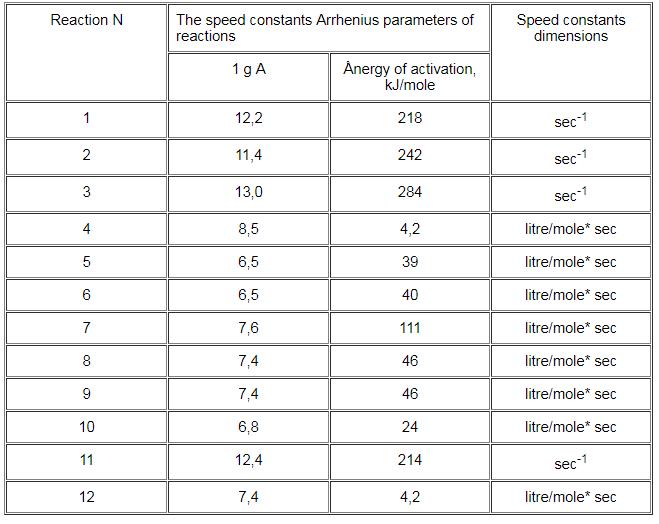
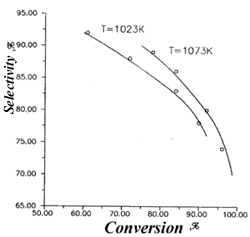 Fig.1. The dependence of trifluorochloroetylene selectivity from 1,1,2-trifluoro-1,2-dichloroethane conversion. | 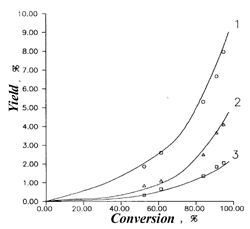 Fig.2.The dependence yields of 1,1,2-trifluoro-1,2-dichloroethane pyrolysis products from conversion. 1-CO, 2-C4F6Cl2, 3- CF2ClH, T=1073 K |
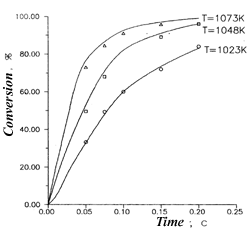 Fig.3.The dependence of 1,1,2-trifluoro-1,2-dichloroethane conversion from retention time. Dilution by water vapor 1:3 | 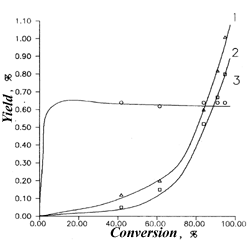 Fig.4.The dependence yields of 1,1,2-trifluoro-1,2-dichloroethane pyrolysis products from conversion.1- C3F4Cl2, 2-C3F5Cl, 3- C2F2Cl2, T=1073 K |
Schemes 1-4 provide some of the experimental data (indicated by points) and design data (lines), obtained with the use of the aforementioned model. A good correspondence of the design and experimental data confirm the adequacy of the process's model.
The maximum selectivity of trifluorochloroethylene has amounted to 80-90%, while at the conversion of 1,1,2-trifluoro-1,2-dichloroethane it reached 85-95%.
Experimental Section.
The analysis of the gas phase thermal decomposition of 1,1,2-trifluoro-1,2-dichloroethane in the presence of aqueous vapor was conducted in the flow reactor, made of the corrosion-resistant alloy XH78T, having the diameter of 4 mm, work surface length – 500 mm. To prevent the cracking of the protective oxide film on the surface of the reactor, the heating and cooling of the reactor were performed in the linear regime with the speed of no more than 100 K per hour. The pyrolysis was carried out in the temperature interval of 973 – 1123 K at the vapor-freon molar ratio 10:1- 2:1.
The chromatographic analysis was executed on the Tsvet-100M chromatographs with the use of the flame-ionization detector and a thermal insulation detector (carrier gas – helium; column length – 3 m; diameter – 2 mm, column temperature – 293-323 K, tricresylphosphate on silochrome – 80).
The identification of the pyrolysis products was performed by the method of chromatomass-spectrometry on the device HP-5995 (the energy of the ionizing electrons – 70 eV, separator temperature – 553 K, ion-source temperature – 423 K).
The mass-spectors of some 1,1,2-trifluoro-1,2-dichloroethane pyrolysis products are provided hereby. The mass-spectors describe the peaks with the respective intensity of more than 2% of the maximum peak, starting with m/e = 35.
(m/e, (I, (I, %))).
Trifluorochloroethylene – 118 (34), 116 (100), 99 (9), 97 (27), 87 (10), 85 (31), 81 (18), 68 (11), 66 (33), 47 (19).
Trifluoroethylene – 82 (82), 64 (3), 63 (100), 62 (4), 51 (50), 50 (7), 44 (6), 43 (4).
1,2-difluorodichloroethylene – 136 (10), 134 (61), 132 (100), 103 (9), 101 (14), 97 (8), 84 (37), 82 (53), 66 (14), 47 (21).
Hexafluoro-1,3-butadiene – 162 (29), 143 (20), 131 (10), 112 (21), 100 (4), 94 (3), 93 (100), 74 (6),
69 (8), 62 (4).
1,1,2,3,4,4-hexafluorodichloro-3-butene – 201 (9), 155 (7), 153 (40), 151 (64), 134 (24), 132 (40), 103 (13), 101 (18), 85 (37), 69 (100).
References
- A.A. Nikolaev, E.P. Goncharov, V.G. Barabanov et al. // The Journal of Applied Chemistry – 1994 – V.67 – issue. 1 – p. 45 – 46
- A.A. Nikolaev, N.V. Mitin, V.G. Barabanov // I Intern. Conf. “Chemistry, Technology and Application of Fluorocompounds in Industry” Abstracts – St. Petersburg – 1994 – p. 63 – 64.
- A.A. Nikolaev, O.V. Rozhdestvenskaya, V.G. Barabanov et // II Intern. Conf. “Chemistry, Technology and Applications of Fluorocompounds” – St. Petersburg – 1997 – p. 156.
- Stephenson I.C., Bialkowski S.E., King D.S. // J. Chem. Phys., 1980, V. 72, N 2, p. 1161 – 1169.
- Hagai K., Katajama M., Nikunia À. // Chem. Phys. Letters, 1979, V. 62, N 3, p. 499 – 502.
- Fields R., Haszeldine R.N. // Y.C. Young. Proc. Chem. Soc., 1966, ¹ 1, p. 22.
- Buxton V.W., Ingram D.W. // J. Chem. Soc., 1952, N 10, p. 3830-3834.
- Solomon W.C., Dee L.A. // J. Org. Chem., 1984, V. 99, N 9, p. 2790-2791.
- Atkinson B., Tsiamis C. // Int. J. Chem. Kinet., 1979, V. 11, N 6, p. 585-593.
- Atkinson B., Stedman M. // J. Chem. Soc., 1962, N 2, p. 512-519.
- P.E. Gill, W. Murroy, M.H. Wright, Practical Optimization,-London, New-York,1981
- A.C.Hindmarsh, A.C. Odepack, A sistematized collection of ODE Solvers,-Lawrence Liwermore Laboratory, Report UCRL-88007, 1982,10p.
Fluorine Notes, 2000, 8, 9-10